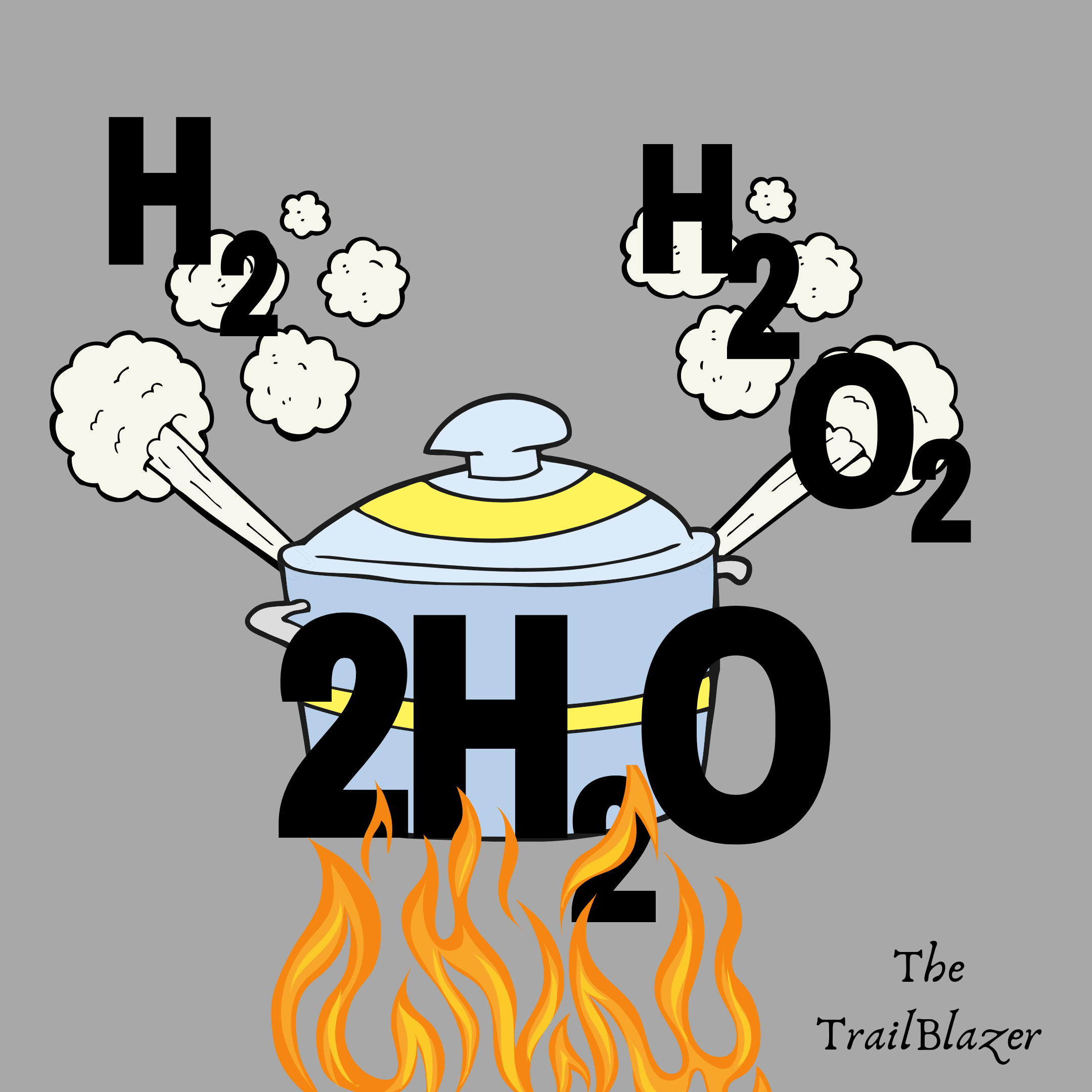
In nature, elements do not usually exist on their individual forms except in few cases. They usually combine with other elements. The combinations are summarised in what are known as the laws of chemical combinations.
- The Law of Conservation of Mass: It states that the total mass of a substance remains unchanged during chemical reaction. This means that in a chemical reaction, the total mass that goes in , equals the total mass that comes out, even when the number and properties of substances change. This law was first stated by Antoine Lavoisier in the 18th century, after series of experiments in combustion. It was discovered that the total amount of matter did not change. Thus, he proposed that matter cannot be created or destroyed in an ordinary chemical reaction. It is also known as the law of indestructibility of matter and basis of writing balanced chemical reaction equations. All quantitative chemistry calculations are based on the law.
Example 1: During one of the Antoine’s experiments, he combusted 5.0g of mercury and produced a calx of mass 7.3g. If calx is made of metal and oxygen , what was the mass of oxygen consumed in his experiment?
Solution:
Total mass in = Total mass out
Mass of metal + mass of oxygen = Mass of calx
5.0g + xg = 7.3g
xg = 7.3g - 5.0g
x = 2.3g
2.3g of oxygen was used.
Exercise:
A chemist heated 1.07g of copper in 3.0g of iodine to produce 3.72g of a white compound. What would be the percentage mass of unused iodine, if all the copper finished?
Leave a comment